Why Are Silicon Nitride Films Grown By LPCVD Denser?
Feb 07, 2025
Leave a message
Why Are Silicon Nitride Films Grown by LPCVD Denser?
0020-40946 CLAMP RING, 8" SNNF, AL
Mechanism of silicon nitride film growth
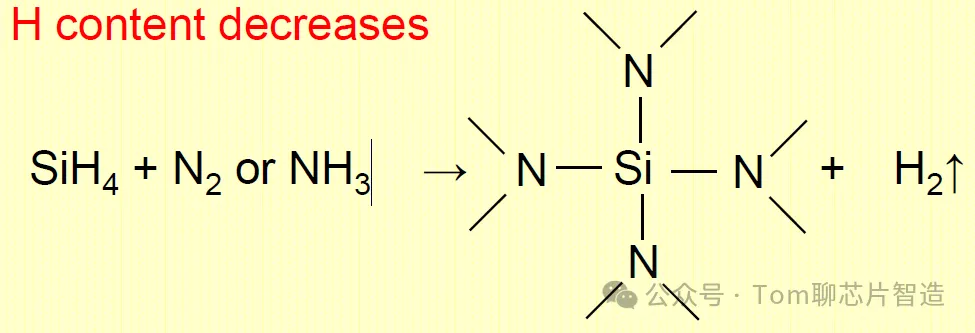
Equation for LPCVD growth:
The equation for PECVD growth:
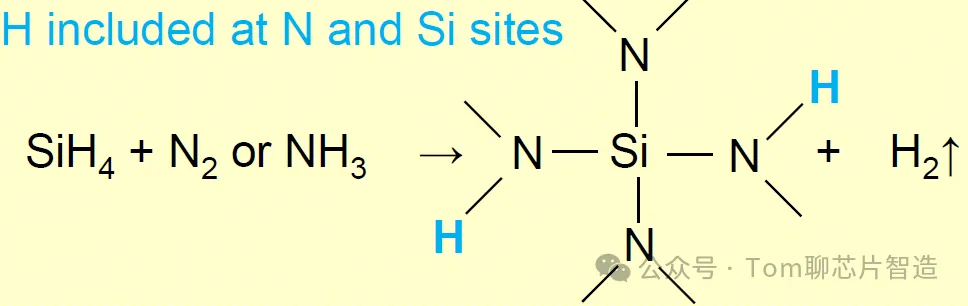
It can be seen that SiH4 provides the Si source and N2 or NH3 provides the N source. However, due to the higher temperature of the LPCVD reaction, hydrogen atoms tend to be removed from the silicon nitride film, so the hydrogen content in the reactants is low. Silicon nitride is mainly composed of silicon and nitrogen. However, the PECVD reaction temperature is low, and the hydrogen atoms can be retained in the film as a by-product of the reaction, occupying the position of N atom and Si atom, making the hydrogen content in the film high, resulting in the resulting film not being dense.
0020-27113 CLAMP RING 6 SMF TI
Why PECVD often uses NH3 as a nitrogen source?
The NH3 molecule contains N-H single bonds, while the N2 molecule contains N≡N triple bonds, and N≡N is more stable and the bond energy is higher, i.e., a higher temperature is required for the reaction to occur. The low N-H bond of NH₃ makes it the preferred nitrogen source for low-temperature PECVD processes.
Send Inquiry


